You will be very surprised why I put CP and autism together in this article but in fact by reading all clinical researches I found that for stem cells therapy protocols for autism studies they are practically same as for CP studies…..
I will start with this powerful video ( Duke):
So I went through the attached Dr K lecture on link (in Israel) and its great. Final found evidence that sex of the donor does not impact success. Some key points:
19:32 mins – starts to talk about autism
21:00 mins – when brain cells are shocked, neurons die unless cord blood is added.
22:07 mins – they took out different cells from the cord blood until they identified CD14 as the only one that stopped the cell death.
27:00 mins – dose is important – high dose showed positive effects.
32:20 mins – 25 million/kg is the higher dose (same dose for the leukemia and CP study).
37:50 mins – With the high dose, MRIs showed improved connections in the brain.
41:00 mins – for genetic conditions, need to do chemo to get the new cells to stay in the brain, this is a serious risk. If genetic issue, use someone else’s cells.
45:00 mins – babies born with autism don’t control which pathways turn on and off, some pathways that are supposed to shut down in early infancy, don’t. This includes microglial activation, which causes sensory issues. (this actually matches Nemechek protocol).
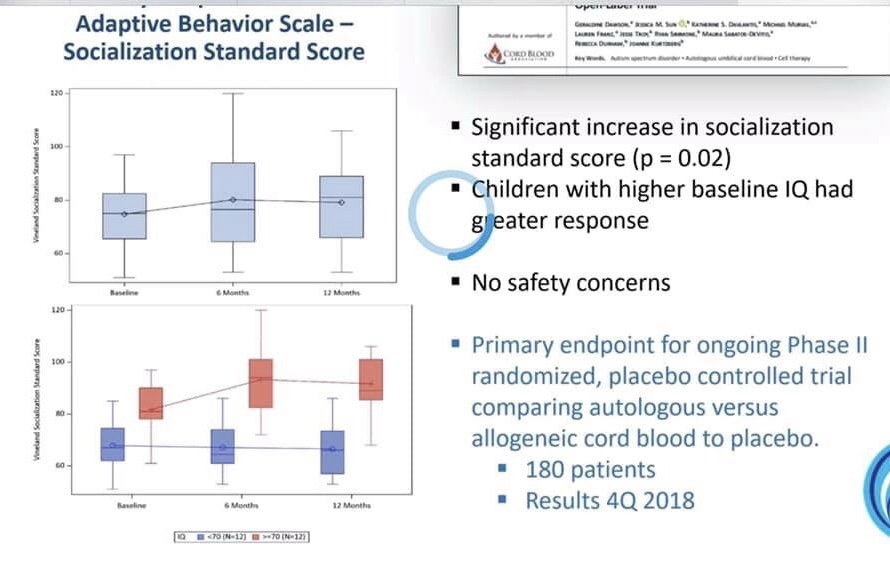
46:30 mins – Vineland would usually go backwards over time as severe kids get older, but during the trial, their scores stayed stable.
51:55 mins – trial due to finish in August, with answers released next fall.
1:09 mins – CD14 cells can’t be expanded. Epilepsy shown no improvement in seizures.
1:12 mins – not know yet whether age impacts success.
1:15mins – the 30% of kids that didn’t show vineland improvement, tended to have lower IQs.
1:17mins – no preference seen whether male or female donors are better.
Cp study 2017 ( Duke)
One more interesting ( but more old video):
donor cord blood from a donor cord blood bank ( only Duke in USA :they just starting first trail with donor cord blood with hla matching….there are some others clinics doing donors cord blood but nobody doing hla matching exept Duke) as well they are doing donors MSC first trail for autism in Duke: This is duke trail autism :
https://clinicaltrials.gov/show/NCT03099239
• Donors MSC from cord tissue ( HLA matching no needed) Duke is doing naw MSC expanded from cord tissue as part of ongoing donor trail for CP : Duke donor trail CP: (cord blood and cord tissue expanded MSC):
So for Duke donor trail CP: (cord blood and cord tissue expanded MSC):
So in this CP trail Duke will decide children’s into 3 groups:
Cord blood group
MSc tissue group here what they will recive:
“the « MSC » arm will receive three hCT-MSC infusions, one each at baseline, three months, and six months” and “Cord Tissue Mesenchymal Stromal Cells
Subjects will receive three intravenous infusions of 2×106/kg human umbilical cord tissue cells (hCT-MSC), manufactured from allogeneic umbilical cord donors”
And Placebo group ( nothing during first year of trail just at the baseline they will recive stem cells treatment )
Contact for Duke :
Duke will do it outside of trials : expanded access or compassionate care.
( but you need to pay for it… Oh and the fee is 15000$) some USA parents got it covered by insurance but not all of them ( diferent insurances may be -don’t know)
Requires :
1) you have cells banked ( own child stem cells from his birth )
2)you have sibling cells banked that HLA matches your child
3) no prior stem cell treatment
4) no ventilator
5) no trach
6)Based on childs weight- Must have a minimum of 25 million cells per Kilogram stored.
7)Duke also turns down if child had immunosuppressive drugs at all.
Process takes anywhere from 6 months to 1 year to get approved. Must turn in a ton of paperwork, records, scans.
3-4 day process ( presence in Duke) 45min IV in arm one injection.
Duke Stem cells contacts:
https://www.dukehealth.org/treatments/cord-blood-transplant
The objective of the study is to enable access to sibling or autologous umbilical cord blood (UCB) infusions for children with various brain disorders.
The use of UCB in this fashion is based on safety and efficacy data from prior and ongoing clinical trials at Duke University Medical Center in over 700 patients with these diagnoses infused with autologous or sibling UCB over the past decade.
Autism Spectrum Disorder
Autism
Cerebral Palsy
Hydrocephalus
Apraxia of Speech
Hypoxia Ischemia, Cerebral
Drowning; Anoxi
Duke:
Adress:
Pediatric Bone and Marrow Transplant Program at Duke
Joanne Kurtzberg , MD
Durham, NC 27710
401-8002
Duke Linet: 919-668-1119
cordbloodtherapyinfo@dm.duke.edu
Bethany Kister RN
Joanne Kurtzberg, MD
Director, Carolinas Cord Blood Bank
Chief Scientific Officer, Robertson Clinical and Translational Cell Therapy Program
Director, Pediatric Blood and Marrow Transplant Program
DUMC Box 3350
2400 Pratt Street Room #9026
Durham, NC 27705
919-668-1119 (phone)
919-668-7161 (fax)
kurtz001@mc.duke.edu
Dr Kurtzberg in Duke university doing just exceptional research for stem cells forCP children’s and for autism ( and if one day it will be recognized by fda it’s because if duke efforts)
Next to watch I will advice you:
Dr. Kurtzberg’s virtual conference at the World Cord-Blood Day 2018:

Extending Cord Blood to Regenerative Therapies for the Brain: Autism, Cerebral Palsy & Stroke: Dr. Joanne Kurtzberg (Duke Department of Pediatrics)
Dr. Kurtzberg’s research has focused on children with selective inborn errors of metabolism. Her work has shown that cord blood cells, administered intravenously after myeloablative therapy engraft in the brain. In addition, DUOC-01, a cord blood derived cellular therapy that promotes myelination, is undergoing testing to augment standard umbilical cord blood treatment in children with leukodystrophies. In this presentation, Dr. Kurtzberg will discuss her on-going research using cord blood to potentially treat autism, cerebral palsy and stroke. She also will explore the potential use of DUOC-1 and highlight the importance of quality control considering the numerous new potential clinical applications of cord blood.
For those that didn’t get the chance to watch it, here’s Dr. Kurtzberg’s virtual conference at the World Cord-Blood Day 2018 that took place on November 15. This link will allow you to bypass the registration process and go directly to the presentation. Once you start watching it, don’t pause or switch apps as you won’t be able to go back (you’ll need to watch the video all over again if you happen to pause.) It takes about 35 minutes to watch the entire presentation. — AND here’s also a link to a separate interview they had with Dr. K :
you can watch other world renowned experts in the field in this link
https://www.worldcordbloodday.org/video-recordings-world-cord-blood-day-2018.html
Very interesting presentation: ( need to register in order to see and listen):

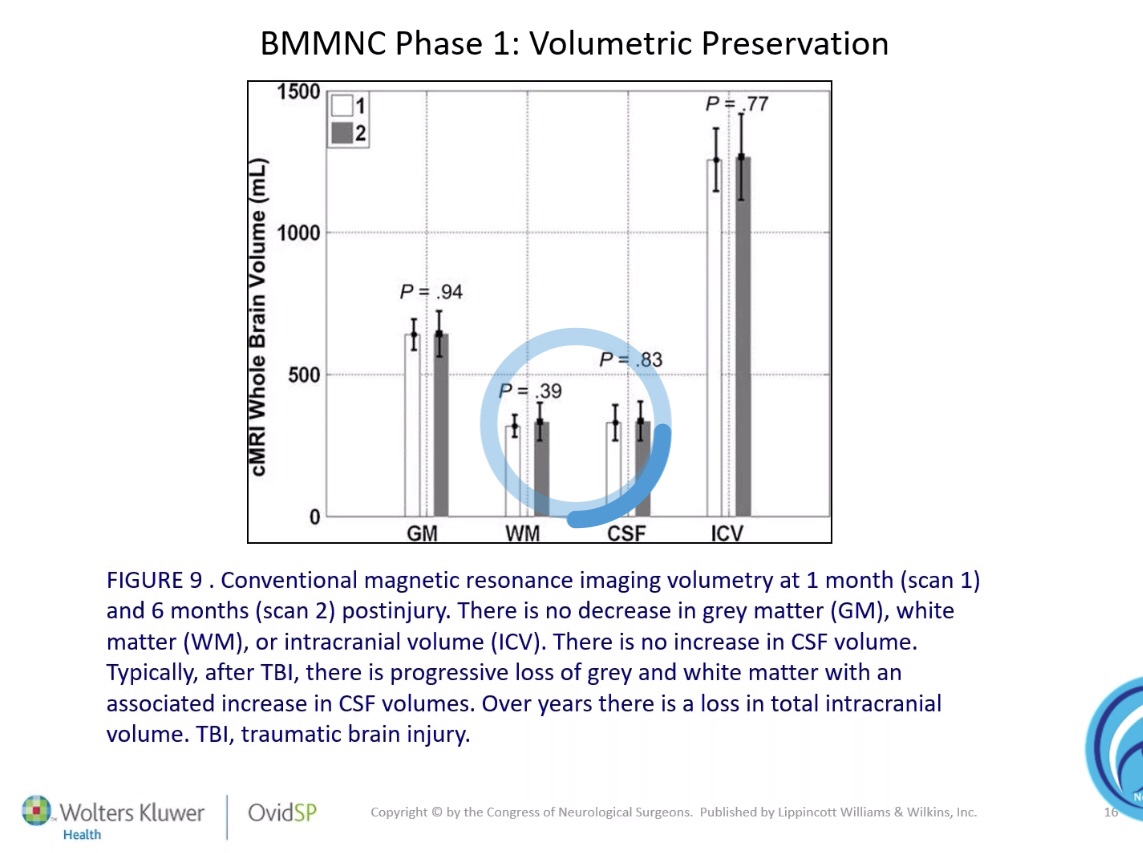
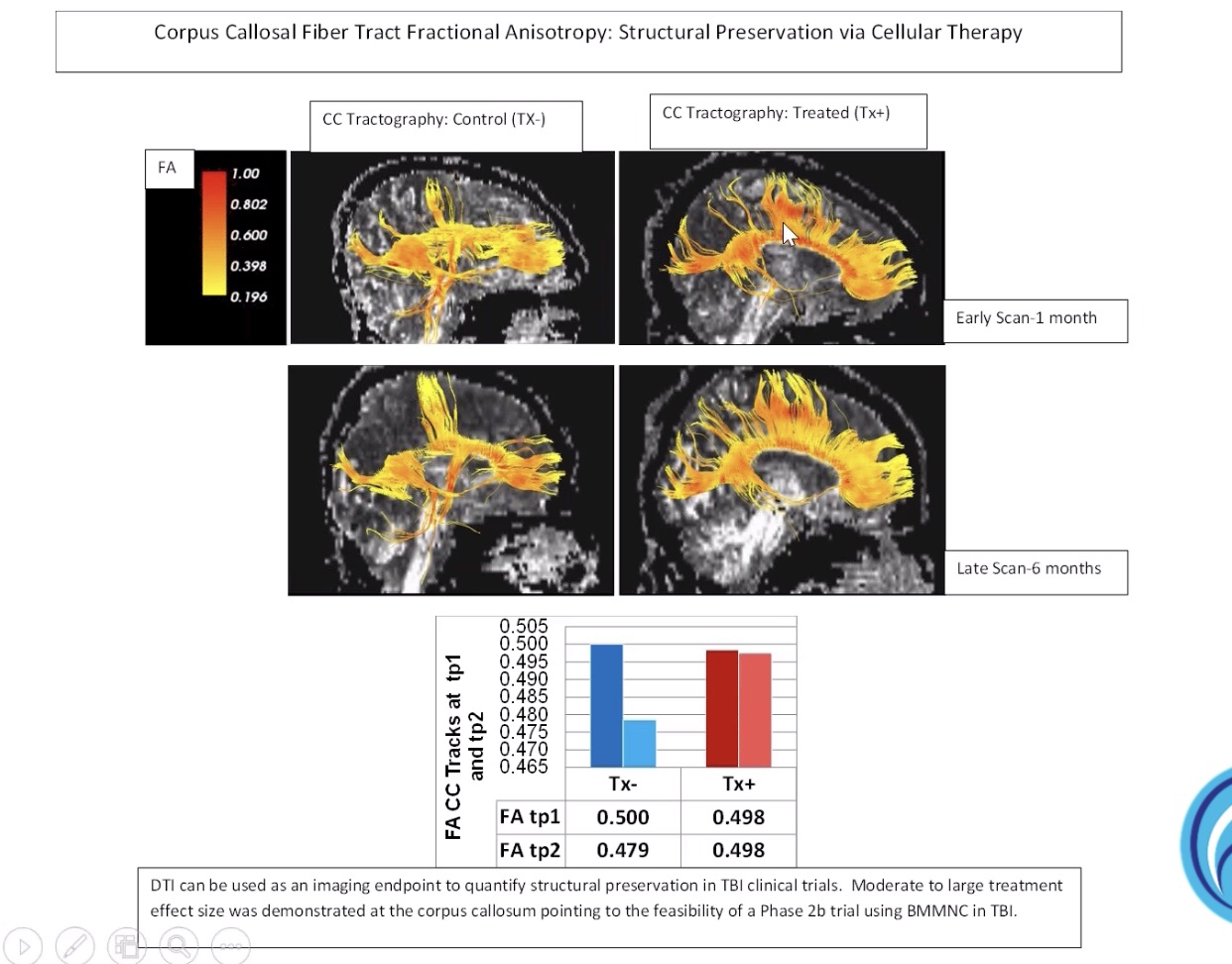
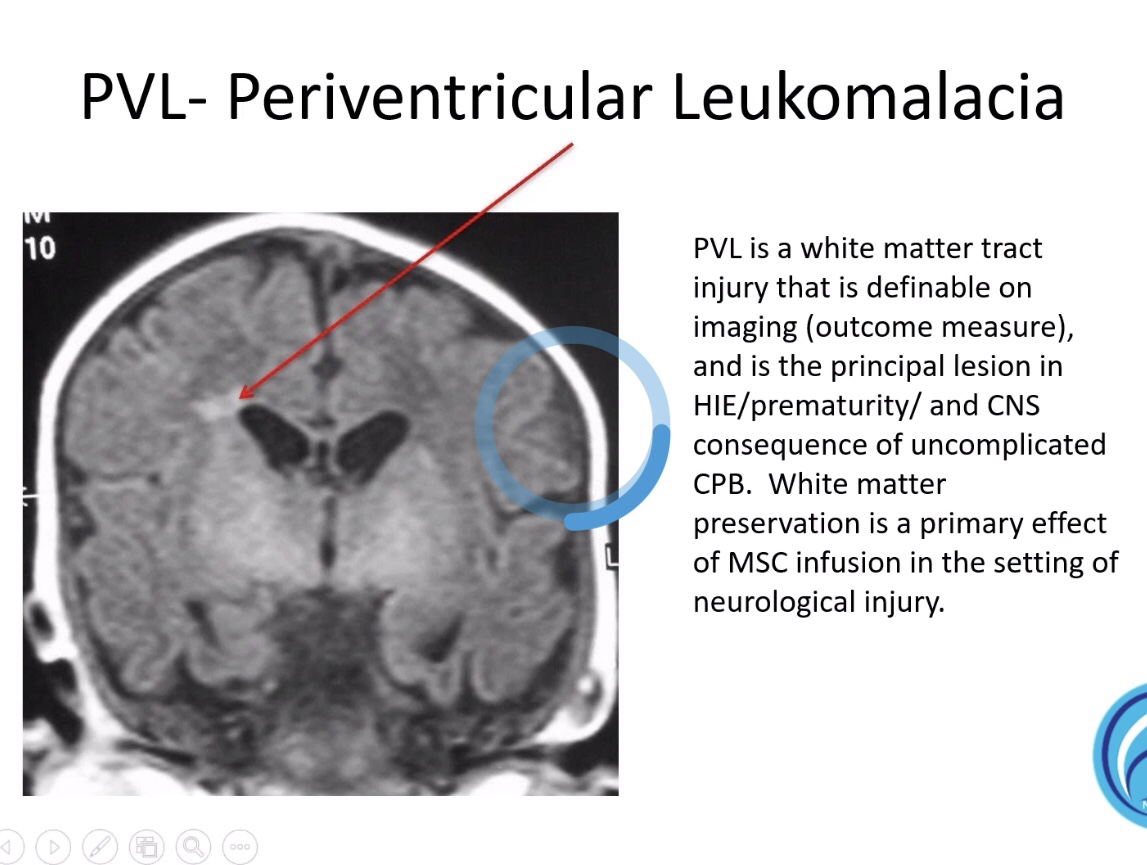
« “Cord Blood for Traumatic Brain Injury / Neurological Injuries” – Dr. Charles Cox : « Neurological injuries have both a primary and secondary component. The secondary component is mediated, in part, by pro-inflammatory cells from the monocyte/macrophage and microglial lineages. Many cellular therapeutics, cord blood in particular can dampen the hyper-inflammatory response to injury that can be deleterious to ultimate repair and recovery. This presentation will highlight some of the potential mechanisms of action that serve as the background for the use of cord blood for these indications. «
French:
Pour ceux qui n’ont pas eu la chance de le voir, voici la conférence virtuelle du Dr. Kurtzberg à la Journée mondiale du selules souches du cordon umbilical 2018, de 15 novembre. Ce lien vous permettra de contourner le processus d’inscription et d’accéder directement à la présentation . Une fois que vous avez commencé à regarder la vidéo, ne mettez pas les applications en pause ni changez d’application, car vous ne pourrez pas revenir en arrière (vous devrez regarder la vidéo à nouveau si vous faites une pause interview commence dans 2-3 minutes de début) Il faut environ 35 minutes pour regarder la présentation entière. Et voici également un lien vers une interview séparée qu’ils ont eue avec le Dr K:
« “Extending Cord Blood to Regenerative Therapies for the Brain: Autism, Cerebral Palsy & Stroke” – Dr. Joanne Kurtzberg (Duke Department of Pediatrics, Marcus Center for Cellular Cures): Dr. Kurtzberg’s research has focused on children with selective inborn errors of metabolism. Her work has shown that cord blood cells, administered intravenously after myeloablative therapy engraft in the brain. In addition, DUOC-01, a cord blood derived cellular therapy that promotes myelination, is undergoing testing to augment standard umbilical cord blood treatment in children with leukodystrophies. In this presentation, Dr. Kurtzberg will discuss her on-going research using cord blood to potentially treat autism, cerebral palsy and stroke. She also will explore the potential use of DUOC-1 and highlight the importance of quality control considering the numerous new potential clinical applications of cord blood.«
